Pink rot of potato is primarily caused by the Oomycete pathogen Phytophthora erythroseptica. Oomycetes are sometimes referred to as water molds due to their strong requirement for free moisture. P. erythroseptica releases swimming spores (called zoospores) under saturated soil conditions.
Pink rot is most effectively managed by utilizing multiple management strategies. Practices that are effective in reducing pink rot severity in potatoes include:
1) Field selection/crop rotation;
2) Adjust soil pH by lime application in low pH soils;
3) Use less-susceptible varieties;
4) Proper irrigation management;
5) Use appropriate fungicides;
6) Avoid "disease-favorable" conditions at harvest;
7) Apply post-harvest fungicides;
8) Reduce tuber pulp temperatures to 55 degrees F or lower;
9) Grade out infected tubers going into storage.
Fungicide Trial
Fungicides with proven efficacy against pink rot in Idaho are represented by four different classes:
1) Mefenoxam/metalaxyl. These include Ridomil Gold products (e.g. Ridomil Gold SL, Ridomil Gold Bravo), Ultra Flourish and MetaStar;
2) Phosphorous acid. Many different phosphorous acid-based fungicides are labeled for use in Idaho on potatoes. Those with published research results include Catamaran, Confine, Phiticide (formerly Crop-phite), Fosphite, Phostrol, Prophyt, Rampart, Resist 57 and Topaz;
3) Cyazofamid (Ranman).
4) Fluopicolide (Presidio).
The relative efficacy of various fungicides has been evaluated at the Miller Research Experiment Farm in Minidoka, Idaho, for the past several years. In 2011, Premier Russet was used for this trial due to its relatively high susceptibility to pink rot. Certified seed was planted with a modified four-row ACME commercial cup-type potato planter (36 inch rows) with 14-inch seed spacing within row, to a depth of 7 inches on May 6. Each plot was four rows wide and 34 feet long with a 6-foot border between plots. Treatments were established in a randomized complete block design with four replications. Fertilizer was applied on May 25 with an air cart spreader and incorporated the same day. Herbicides were applied on May 27 with a Melroe Spray Coupe and incorporated with irrigation.
In-furrow application was made at planting on May 6. Products were applied with TeeJet SS8001E nozzles mounted on the planter. Nozzles (one per row) were positioned to spray the soil as it was being turned into the furrow. Products were applied using 9.1 gallons of spray mix-per-acre. Red Ball flow indicators were used to monitor the output for each nozzle and help ensure accurate and uniform spray delivery for each row in each plot. The spray lines were flushed between each plot. Nozzles were positioned 8 inches above the opened furrow and tilted so the spray was applied to the soil as it was turned over and around the seed piece.
Foliar applications were applied using a ground-based plot sprayer (a small self-propelled tractor with a hydrostatic drive) on the dates listed in Table 1. The first application was made when the largest tubers were dime size. The spray boom consisted of eight Teejet XR 11002 vs. flat fan nozzles spaced 18 inches apart. Applications were made using 20-psi pressure to apply 16.5 or 17.3 gallons of spray mix per acre. The boom was positioned about 18 inches above the crop canopy. 
Pink rot incidence was evaluated using the same technique as outlined for the irrigation trial above. Pink rot incidence was present at a relatively low level in the untreated check (2.3 percent) and none of the treatments significantly reduced disease (Table 3). Our experience has been that it is difficult to detect an impact of fungicides when disease pressure is low.
Isolates of Phytophthora erythroseptica were obtained from infected tubers from the untreated check. All isolates were highly sensitive to mefenoxam. Resistance to mefenoxam has been found in the field where this trial was conducted in the past. However, none were recovered this fall from the tubers tested. 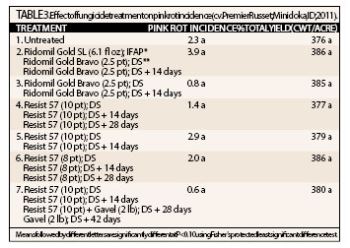
Historically, programs with mefenoxam/metalaxyl have been the most effective. Foliar applications of mefenoxam/metalaxyl have been more consistent than in-furrow applications. Where pathogen resistance to mefenoxam/metalaxyl has been found, applications of phosphorous acid have been the most effective alternative. Treatment 4 in Table 3 above has been the most consistent program for pink rot control with phosphorous acid.
Phytotoxicity has been observed with some foliar phosphorous acid applications. We initiated a trial where phosphorous acid was applied with a simulated aerial application (six gallons per acre carrier volume applied by ground). Applications were made either in the morning (about 6 a.m.) or in the late afternoon (about 6 p.m.). Leaf burn was only observed with applications made in the early morning. Based on this observation, it may be possible that phytotoxicity is some way associated with applications made during period of high relative humidity. Additional work is needed to confirm this observation.
Post-Harvest Applications
Post-harvest application of phosphorous acid has also been effective in managing pink rot. The post-harvest application will not cure tubers that are already infected at harvest, but it will protect healthy tubers from becoming infected as healthy and infected tubers come in contact during the handling procedures at harvest.
Applications of 12.8 fl oz of phosphorous acid in a carrier volume of 0.5 gallons of water per ton of tubers has been the most effective for controlling pink rot development in storage. (This application is also effective in controlling late blight in storage.)
Phosphorous acid fungicides with post-harvest research efficacy data include Confine, Fosphite, Phiticide, Phostrol, Prophyt and Resist 57.
Some concern has been raised about making post-harvest applications to a potato crop destined for use as seed the following year. Research at the University of Idaho has shown that post-harvest applications of phosphorous acid are not detrimental to potato seed performance. However, it is not recommended that phosphorous acid be applied to short dormancy varieties that may sprout when being harvested. Additionally, phosphorous acid is not recommended as a seed treatment. It is not effective when used that way, and we have observed delayed emergence. No adverse effects have been observed with processing quality with respect to glucose and sucrose content and fry color.
Application of phosphorous acid through the ventilation system has been evaluated at the University of Idaho. Late blight incidence was reduced from 37 percent in the untreated check to 8 percent with an application of Resist 57. It has proven difficult to keep phosphorous acid suspended in the ventilation system. Additional work is needed in this area. An attempt was made to create a thermal aerosol with phosphorous acid. This resulted in the generation of the toxic and flammable phosphine gas.
Growers worried about controlling pink rot can utilize irrigation management and fungicides (among other management practices) to help minimize the impact of pink rot. Data from 2010 and 2011 show that irrigating more frequently with less water results in less pink rot than irrigating less frequently with more water for a given amount of water. Fungicides did not show a significant reduction in disease under low disease pressure in 2011. Post-harvest application of phosphorous acid is an effective tool for reducing pathogen spread at harvest.