Misdiagnosing a disease can be very costly to a grower if unnecessary fungicide applications or other control measures are implemented. There are many real-life examples where proper identification of a disease was not made until after avoidable inputs were applied, management changed or rumors started.
During the growing season, there may be times when there is a delay in identification of samples submitted to the University of Idaho due to personnel being out of the office or the diagnostic technique taking time to run.
It would be beneficial to growers, field representatives and extension personnel to have rapid and simple disease identification test kits available to make accurate initial disease diagnoses in the field.
These test kits can also be helpful in eliminating disease misdiagnoses and determining whether or not the problem is physiological or chemical and not due to a pathogen at all.
A program was developed to evaluate the use of commercially available pathogen test kits and determine the accuracy of each test kit. Multiple test kits for numerous diseases were used, assessed and compared to results from traditional diagnostic methods.
Diagnostic test kits were purchased from four major companies (Table 1) and sent to potato researchers throughout Idaho. Samples collected in fields or grower samples brought to labs were tested with the kits and pathogens were identified using traditional laboratory analysis techniques to verify accuracy of the kits (eg. RT-PCR, ELISA, plating, etc.).
"Ease of use" surveys were filled out for each kit. A result reported as a "false positive" was defined as a test kit result of positive but the traditional laboratory method result was negative. Conversely, a "false negative" result occurred when a test kit indicated a negative result and the traditional laboratory test was positive.
Viruses (PVY, PVY strains, PLRV, AMV)
Potato leaf samples collected in fields or submitted grower samples were tested with the kits and analyzed for virus verification using conventional ELISA and RT-PCR protocols. Both PVY symptomatic and non-symptomatic leaf samples were collected and tested. Approximately 115 samples were tested using 154 PVY kits and 44 samples tested using 60 PVY"N" kits. PVY test kits from the four companies were 94 to 97 percent accurate. One company's PVY test kits had 3 percent false negative and 5 percent false positive results. The remaining three companies' test kits showed 5 to 9 percent false negative and 0 percent false positive tests. Results indicate that PVY test kits displayed broad reactivity and correctly detected PVY"O", PVY"N:O", or PVY"NTN". The PVY"N" test kits were 90 percent accurate overall and gave one false negative and two false positives. The PVY"N" test kits showed a false positive for PVY"N" when the laboratory result was PVY"O". Results indicated the kits tested were reliable and accurate, but some limitations were apparent. In this study, using a general PVY test kit identified infection by common strains of PVY that are observed in Idaho and the Pacific Northwest. 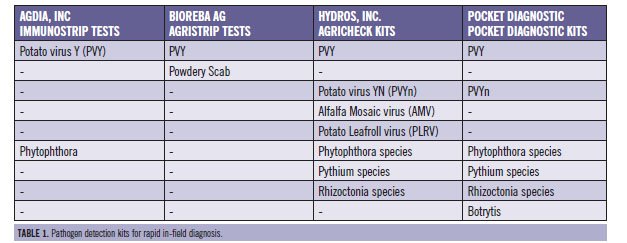
A limited number of leaf samples were tested for PLRV due to the low levels of the virus in commercially grown potatoes. The test kit used failed to identify the infected plant. No positive samples of alfalfa mosaic virus were found and all test kits used correctly identified the samples as negative. Additional testing is required of the PLRV and AMV test kits to verify the usefulness of these virus test kits.
Phytopthora (late blight and pink rot)
There was limited opportunity to test pathogen kits on positive foliar late blight samples. Ranger Russet foliar late blight samples were tested in Bonner's Ferry, Idaho, and the Agdia Phytopthora kit correctly identified the positive and negative samples. On the same late blight samples, the Hydros phytopthora kit gave two negative results and one was a false negative. Both test kits correctly identified both positive and negative late blight tuber samples. Several test kits were used successfully on grower samples, resulting in a negative answer for foliar late blight.
Obtaining the correct positive results using the phytopthora kits for pink rot infection of stolons, stems or tubers proved to be a challenge. One researcher noted a correct result when the sample was agitated longer than the kit instructions specified. Otherwise the test kits were fairly effective at identifying pink rot-infected tubers even when the tuber was succumbing to secondary invasion by bacterial soft rot although the indicator line was often fairly faint.
Botrytis (gray mold)
Grower samples with dark foliar lesions were tested for botrytis. All seven of these samples tested negative for botrytis and were confirmed negative by a pathologist. The samples' symptoms were physiological problems, not botrytis. Additional testing is required of the botrytis test kits to verify the usefulness of the kit.
Rhizoctonia and Powdery scab
Test kits had limited success correctly diagnosing rhizoctonia on stem, stolon and tuber tissue.
Several samples for powdery scab were tested with high accuracy and results would recommend using this test kit to verify if the lesions are powdery scab or common scab. 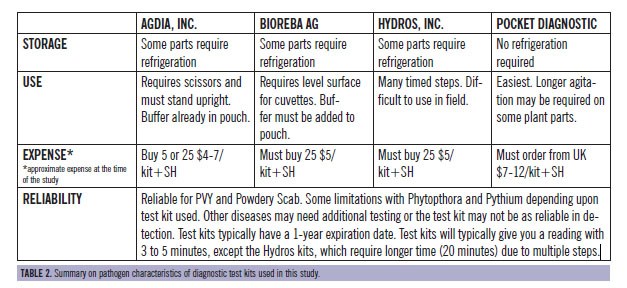
Pythium
Pathogen test kits were used on tuber samples from a Pythium disease inoculation study. The Pocket Diagnostic test kits were accurate in diagnosing tuber infection. Even tubers with significant secondary bacterial soft rot tested positive for Pythium, indicting the initial source of the breakdown. This test kit will be helpful in identifying the causal factor in tuber breakdown in storage.
Results indicated the kits tested were reliable and accurate, but some limitations were apparent. Overall, the use of disease diagnostic test kits will provide an additional tool for greater accuracy, efficiency and sustainability. Most of these test kits can be used in the field, storage or office. A laboratory is not required. In order to use this additional diagnostic tool to your advantage, you must select a test kit based upon the symptoms since each test kit is specific to a particular pathogen. Due to the potential for false negatives and/or false positives, it is imperative to have samples subsequently verified by the University of Idaho or an independent laboratory. This additional contact will also provide management suggestions or input on the situation.